 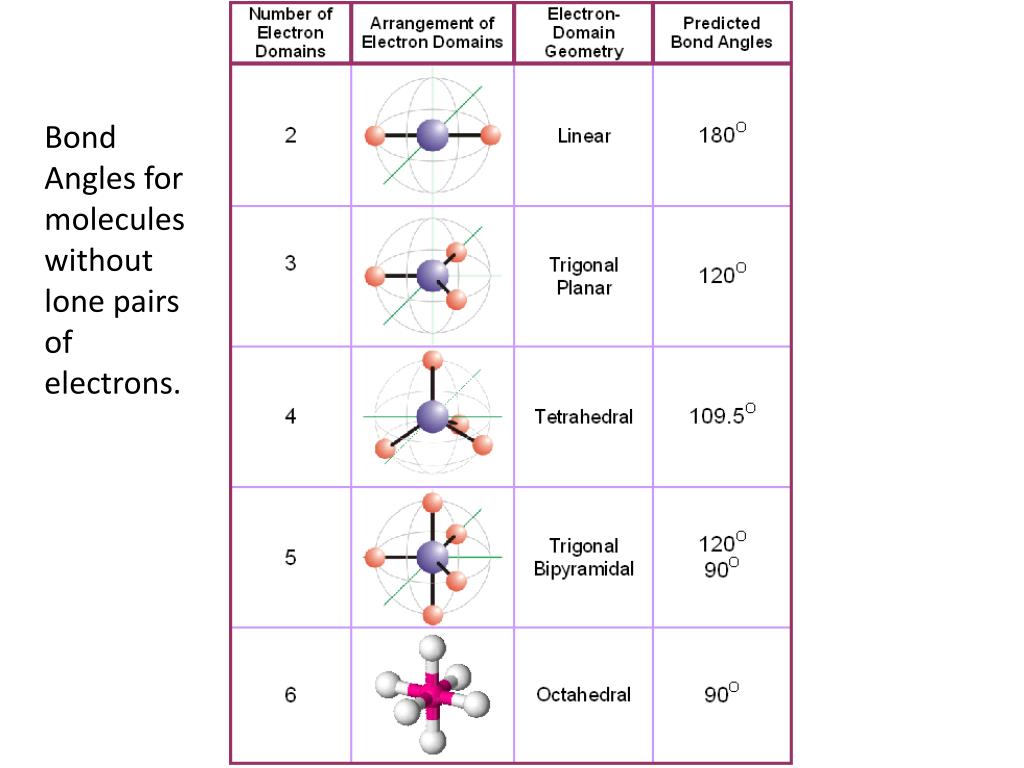
In what state of hybridization the compound is present, can be found by knowing its Steric number. Hybridization in simple terms means the distribution of electrons in different energy orbitals to get an idea about the compound and its properties.ĭuring hybridization, the properties of the compound can be altered minutely or drastically depending on the compound and conditions. You cannot complete your understanding of a compound if you are not thorough with the basic concept and idea of hybridization. Hybridization is a very common term and concept when we learn about the bonding in compounds. Moving to the next section, we will be learning about the hybridization of CCl4 Look at the diagram below to get a visual representation of what we just learned. This takes us to the conclusion that there are 6 valence electrons on each of the Chlorine atoms after bonds are formed. These 24 are non-bonding electrons and they form 12 lone pairs in the CCl4 compound. Now, the remaining number of electrons is: Thus, 4 electrons of Carbon and 4 electrons of Chlorine are used in bond formation, which totals as 8 electrons. Here all four electrons of Carbon participate in bond formation with one electron of all four Chlorine atoms. The final sum of C and Cl electrons thus comes up to, So, total number of Valence Electrons is, We will be needing to add all the electrons that are bonded and those which are lone to get the number of Valence electrons. To understand this better, let us calculate the Valence electrons of CCl4. The other electrons that are left form a lone pair. In every compound there is a central atom, in the case of CCl4, the central atom is Carbon to which atoms of Chlorine are bonded or attached.Īll the electrons that are shared between C and Cl form a bond. Let us not waste time in understanding the structure of CCl4. Now as you know the concept of how the Lewis structure is constructed and what is its importance. Let us quickly hop on to the next segment which is understanding the Lewis structure of CCl4. According to which there should be 8 electrons in the outer shell of an atom. 
The bonds and lone pairs are collectively known as Valence electrons.įor any compound to be stable, the octet rule has to be followed. Lone electrons are represented as dots in the Lewis structure, whereas, bonds are represented as a single line in the structure.Īll the electrons that take part in forming bonds are known as bonding pairs of electrons.Īnd the electrons that do not take part in forming bonds are known as non-bonding pairs of electrons. This nomenclature is there because of the bond representation that takes place within the compound. The Lewis structures are also known as Lewis dot diagrams or electron dot diagrams. In a Nutshell How to Draw Lewis Structureĭrawing any compounds Lewis Structure is paramount due to one main reason- “you understand the properties of compound better”. Now let’s move into the study of its structure and other details, starting from its Lewis structure. These are some of the basic properties of Carbon tetrachloride. If the compound is consumed or exposed to humans in large or continuous motion then the person or group of people can suffer from kidney or liver damage. If any human inhales CCl4 compound (short-term) or gets orally exposed to it then they can feel headache, lethargy, weakness, and nausea. CCl4 will release toxic fumes like carbon monoxide. The boiling point of CCl4 is 76.8 degrees Celcius and its melting point is -23.0 degrees Celcius. The liquid is not soluble in water and is non-combustible. The compound is clear and very stable in nature.ĬCl4 is also named carbon chloride, methane tetrachloride, benziform, and more. Initially, the compound had everyday applications but considering its harm to humans the chemical is now banned from use. In its natural state, it is a colorless liquid chemical with a little sweet smell like ether. Carbon Tetrachloride is a chemical that is produced and is not available naturally. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
